Homework for Chapter 6 - Stability and Cloud Development
SHOW ALL WORK , CIRCLE THE CORRECT ANSWER, PLEASE BE NEAT AND STAPLE YOUR HOMEWORK!
Follow the Problem solving steps discussed in class
1. Given the following surface conditions:
a. T = 25 degrees C, Td = 22 degrees C
b. T = 35 degrees C, Td = 17 degrees C
c. T = 23 degrees C, Td = 9 degrees C
d. T = 15 degrees C, Td = 0 degrees C
e. T = 40 degrees C, Td = 6 degrees C
For which conditions would one see the highest cloud base and the lowest? What would the cloud base height be? Calculate cloud base using line slope formulas.
2. If the height of a cumulus cloud is 1000m above the surface, and the dew point as the earth's surface beneath the cloud is 20 ºC, determine the air temperature at the earth's surface beneath the cloud.
3. Suppose the air pressure outside a conventional jet airliner flying at an altitude of 10 km is 250 mb. Further, supose the air inside the aircraft is pressurized to 1000mb. If the outside air temperature is -50 ºC, what would be the temperature of this air if brought inside the aircraft and compressed at the dry adiabatic rate to a pressure of 1000mb (assume that a pressure of 1000mb is equivalent to an altitude of 0 m).
4. Print out this SkewT diagram.
EXTRA CREDIT: The buoyancy force that a parcel of air experiences is created when a density difference between the environmental air and the parcel exists. Per unit mass, the buoyancy force may be expressed as:
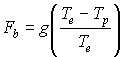
where g is the acceleration due to gravity, Te and Tp are the environmental and parcel temperatures, respectively. Show that the buoyancy force is positive (directed upward) when the density of the parcel is less than the density of the environment. (HINT: Use the ideal gas law)